The Trending With Impact series highlights Aging (Aging-US) publications that attract higher visibility among readers around the world online, in the news, and on social media—beyond normal readership levels. Look for future science news about the latest trending publications here, and at Aging-US.com.
—
What if drugs designed to treat conditions such as diabetes, osteoporosis and rheumatoid arthritis could at the same time provide patients with anti-aging benefits? On March 29, 2022, researchers—from Insilico Medicine, University of Chicago, George Mason University, University of Liverpool, and Buck Institute for Research on Aging—released a new study on the cover of Aging (Aging-US) Volume 14, Issue 6, about Insilico’s next-generation artificial intelligence (AI)-powered discovery software, called the PandaOmics platform. Their trending research paper is entitled, “Hallmarks of aging-based dual-purpose disease and age-associated targets predicted using PandaOmics AI-powered discovery engine.”
PandaOmics
The PandaOmics platform is a software based on an enormous database of research that is continuously being updated and refined. The database consists of over 1,500 diseases and 10,000 disease subtypes, approximately 1.9 trillion data points derived from over 10 million samples with microarrays, RNA sequencing, proteomes, methylomes, and other data types, 1.3 million drug compounds and biologics, and information embedded from over 40 million text-based sources and documents. This evolving omics database is then used to inform intricate AI algorithms designed to identify patterns within the data.
The Hallmarks of Aging
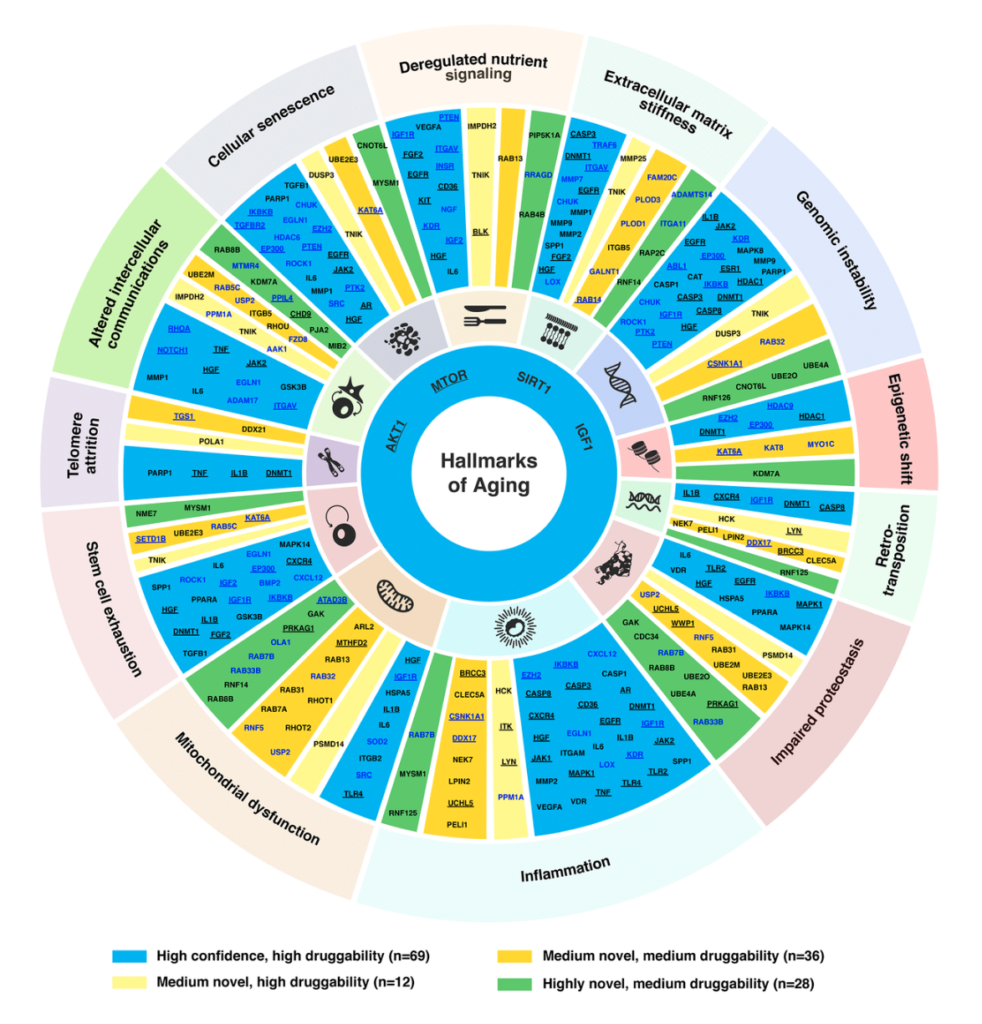
While the underlying molecular mechanisms of aging are still technically in debate, researchers have basically agreed upon a consistent series of biochemical changes that have been identified in the aging process, known as the hallmarks of aging. There are nine classic hallmarks of aging, as well as three newer additions, which include: 1) altered intercellular communications, 2) cellular senescence, 3) deregulated nutrient signaling, 4) epigenetic shift, 5) genomic instability, 6) impaired proteostasis, 7) mitochondrial dysfunction, 8) stem cell exhaustion, 9) telomere attrition, 10) inflammation, 11) extracellular matrix stiffness, and 12) retrotransposons.
Researchers have observed that substantial overlap exists between genes involved in the hallmarks of aging and in age-associated diseases (AADs). The goal of this study was to use the PandaOmics platform and the hallmarks of aging to identify dual-purpose drug targets that can be used to treat both aging and AADs.
“Hence, identifying potential targets that are implicated in multiple age-associated diseases, and also play a role in the basic biology of aging, may have substantial benefits.”
The Study
In this study, the researchers used the PandaOmics platform to generate a list of promising new or traditional aging-associated targets that may be used for drug discovery and repurposing. The team started by investigating genes that are dysregulated in multiple aging-associated diseases, as well as in aging itself. The researchers decided only to analyze 33 diseases. This decision was based on whether or not age is a strong risk factor for the disease’s onset, if there are strong confounding factors and on the availability of public datasets. Cancers and cardiovascular diseases were excluded from this list of selected diseases (this list can be found in the study).
The selected diseases were separated into AADs (n=14) and non-age-associated diseases (NAADs) (n=19). The team then programmed the PandaOmics platform to identify aging-associated patterns by prioritizing the top dysregulated genes in these diseases based on their involvement in the hallmarks of aging. The researchers compared the top-AAD and -NAAD genes and identified 145 overlapping common targets.
Results
“In this study, we used a variety of target identification and prioritization techniques offered by the AI-powered PandaOmics platform, to propose a list of promising novel aging-associated targets that may be used for drug discovery. We also propose a list of more classical targets that may be used for drug repurposing within each hallmark of aging.”
They found that most aging-associated targets were not specific to a single hallmark but were instead involved in multiple hallmarks. The team also found that most of the top targets played a role in the 10) inflammation and 11) extracellular matrix stiffness hallmarks of aging. Four targets were connected to all 12 hallmarks; these targets were AKT1, MTOR, SIRT1, and IGF1. Primary conclusions drawn from the study were that the hallmarks of aging are implicated in multiple AADs and NAADs, and that these hallmarks can be used to identify aging-associated targets for drug discovery and repurposing.
Conclusion
The researchers were forthcoming about limitations in this study. Nevertheless, this exciting research provides valuable insight into the use of AI-powered discovery engines to uncover novel aging-associated targets for drug discovery. The PandaOmics platform is a valuable resource for aging researchers and offers the potential to identify new or traditional targets for the treatment of aging and age-related diseases.
“In conclusion, we successfully established an approach to identify potential dual-purpose targets for aging and AADs, enabling biologists and clinicians to further investigate their therapeutic potential in a cost-saving and time-efficient manner for drug discovery. These promising results underscore the ability of PandaOmics to identify novel targets not only for specific disorders, but across multiple types of diseases.”
Click here to read the full cover paper published in Aging (Aging-US).
AGING (AGING-US) VIDEOS: YouTube | LabTube | Aging-US.com
—
Aging (Aging-US) is an open-access journal that publishes research papers bi-monthly in all fields of aging research. These papers are available at no cost to readers on Aging-us.com. Open-access journals have the power to benefit humanity from the inside out by rapidly disseminating information that may be freely shared with researchers, colleagues, family, and friends around the world.
For media inquiries, please contact [email protected].

