The Trending with Impact series highlights Aging publications that attract higher visibility among readers around the world online, in the news, and on social media—beyond normal readership levels. Look for future science news about the latest trending publications here, and at Aging-US.com.
—
From birth to advanced age, chemical changes occur that affect the genetic material in human cells comprising chromosomes—known as chromatin. These nongenetic changes, otherwise referred to as epigenetic aging, impact transcriptional programs and accumulate with age. From growth and development to adulthood, these changes can denote shifting epigenetic contexts.
“Shifting epigenetic contexts influence regulatory variation and disease risk”
There is a considerable amount of evidence that suggests a causal relationship between changes in epigenetic state and cell aging. Curiously, researchers have repeatedly observed similar epigenetic changes occurring across different cell and tissue types, throughout different stages of life. These potentially synchronized changes may implicate mechanisms of the aging process.
“Together, these findings suggest that a central trajectory for epigenetic state that reflects innate aging processes may exist [20], upon which extrinsic and cell-type effects are layered.”
In 2021, researchers from Harvard University and the Broad Institute wrote a theory article that was published in Aging’s Volume 13, Issue 12, and entitled, “Shifting epigenetic contexts influence regulatory variation and disease risk.” The authors described common epigenetic trends throughout human growth, development, and aging. They also aimed to show how changing epigenetic contexts may influence the behavior of evolutionary forces and risk of genetic disease.
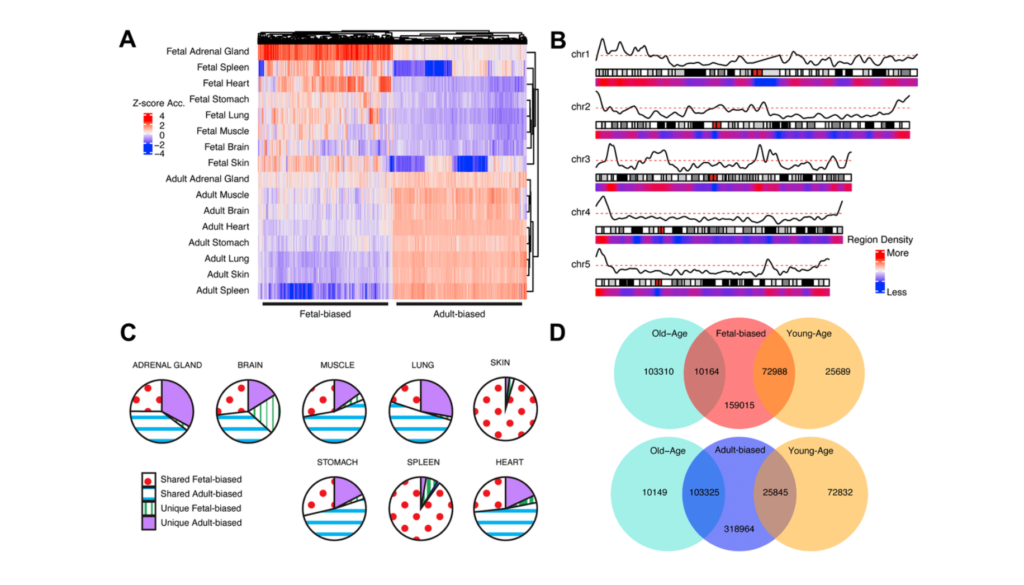
Fetal to Adult Epigenetic Shifts
Some epigenetically-regulated biological pathways involved in development (such as Wnt) eventually play a role in adult tissue homeostasis. These pathways are also implicated in age-related tissue decline. Researchers suggest that this indicates a molecular link between the processes that mediate growth and aging.
“Epigenetic marks established during development can persist into adulthood [9], but they do so in the context of shifts in epigenetic states (see below) as tissues transition into their adult forms and functions.”
In order to better understand the contribution of epigenetic changes to disease and aging, the researchers in this study point out that it is important to understand the developmental changes that occur between fetal and adult tissues, and their interaction with epigenetic aging. Characterizing these epigenetic trends and examining their potential interaction with later-in-life epigenetic aging were main goals of this study.
“Furthermore, these fetal to adult epigenetic shifts can be compounded by additional modifications through aging-associated epigenetic changes.”
To characterize epigenetic trends, the researchers defined genomic regions where chromatin accessibility consistently shifts. The authors note that they used an accessibility-based definition of epigenetic context, and that there are other marks of epigenetic changes (e.g. methylation, and etc.) that are not captured by this definition. Chromatin can be broadly classified in one of two epigenetic states: activating or repressing modifications. These states refer to chromatin accessibility and the increased or decreased ability of DNA to access gene-regulatory machinery, such as transcription factors.
Conclusion
The researchers utilized genome-wide association study (GWAS) datasets to find that gene variants in adult tissues gaining nearby accessibility have stronger associations across a number of aging-related diseases, including neoplasms, arthritis, and atherosclerosis.
“In other words, it is the change in epigenetic context that modifies the regulatory potential of these variants, and this has direct impacts on individual associations with multiple diseases.”
Among their many findings, the researchers explain that the regulatory sequences which are most active during development are subject to strong negative selection later in life.
“We utilize our findings to propose a model for how evolutionary forces may have acted at these loci in humans, and how these forces in turn influence the distribution of mutations conferring heritable disease risk across a number of age-associated pathologies.”
Click here to read the full theory article, published by Aging.
WATCH: MORE AGING VIDEOS ON LABTUBE
—
Aging is an open-access journal that publishes research papers monthly in all fields of aging research and other topics. These papers are available to read at no cost to readers on Aging-us.com. Open-access journals offer information that has the potential to benefit our societies from the inside out and may be shared with friends, neighbors, colleagues, and other researchers, far and wide.
For media inquiries, please contact [email protected].