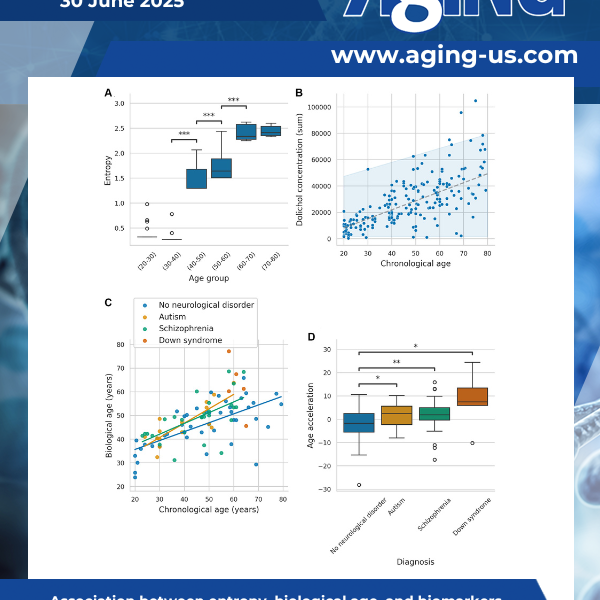
This study introduces DoliClock, a lipid-based biological aging clock designed to predict the age of the prefrontal cortex using post-mortem lipidomic data. Significant age acceleration was observed in autism, schizophrenia, and Down syndrome.
Aging (Aging-US) Research
As we age, our brains become more sensitive to stress and disease. A recent study sheds light on a lesser-known risk: reduced oxygen levels.
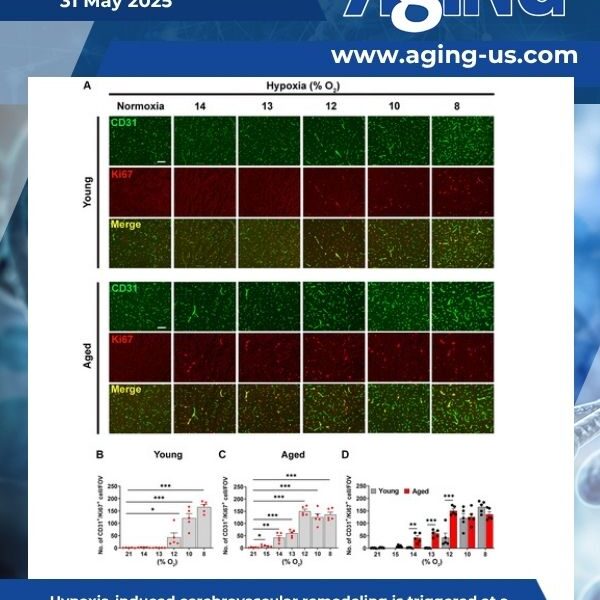
Chronic mild hypoxia (CMH; 8% O2) triggers transient blood-brain barrier (BBB) disruption, an effect greatly increased with age. As BBB disruption predisposes to neuronal death and cognitive decline, here we defined the hypoxic thresholds that trigger BBB breakdown in young and aged mice, and then defined the age at which hypoxia-induced BBB disruption significantly increases.

Werner syndrome is a rare condition marked by accelerated aging. A recent study, featured as the cover paper in Aging (Aging-US), Volume 17, Issue 4, led by researchers at the University of Oslo and international collaborators, suggests that nicotinamide adenine dinucleotide (NAD+), a vital molecule involved in cellular energy production, may be key to understanding this disease and developing future strategies to manage it.
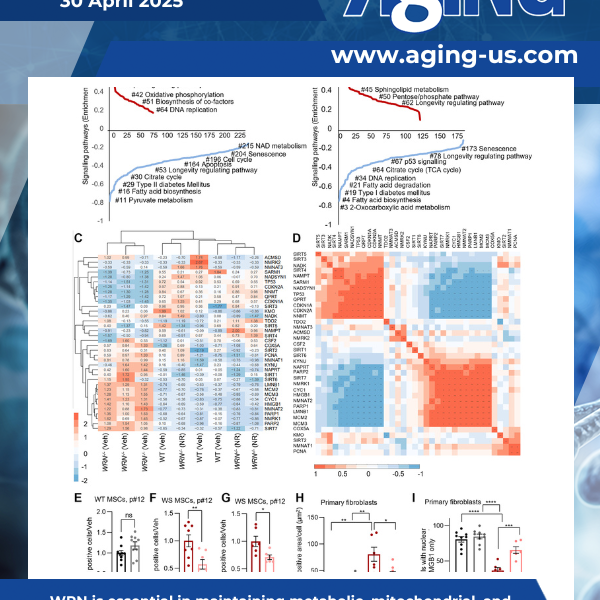
Werner syndrome (WS), caused by mutations in the RecQ helicase WERNER (WRN) gene, is a classical accelerated aging disease with patients suffering from several metabolic dysfunctions without a cure.
Could a class of drugs that clear aging cells also help treat Alzheimer’s disease? A recent study, featured as the cover for Aging (Volume 17, Issue 3), titled “Differential senolytic inhibition of normal versus Aβ-associated cholinesterases: implications in aging and Alzheimer’s disease,” suggests they might—and with remarkable precision.
Radiation therapy or radiotherapy, is a common treatment for cancer, but its effectiveness differs across patients. A recent study published as the cover for Volume 17, Issue 2 of Aging explored why this happens. The findings provide valuable insights, particularly for brain cancers like glioblastoma (GBM) and low-grade gliomas (LGG).
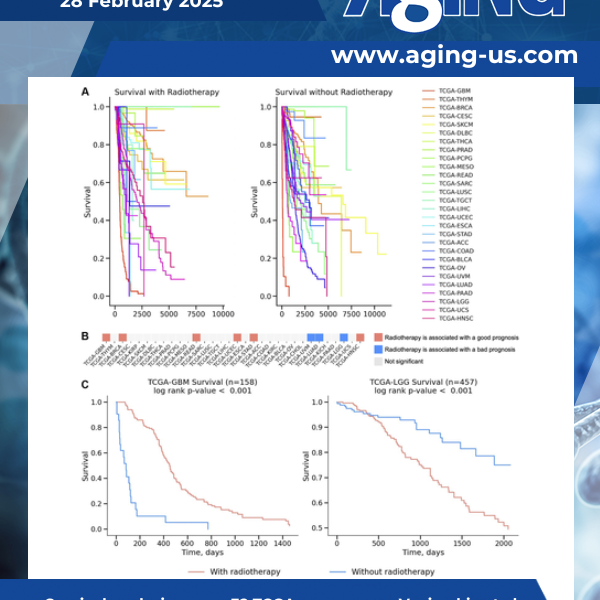
Radiotherapy is a crucial treatment option for various cancers. However, the results of radiotherapy can vary widely across different cancer types and even among patients with the same type of cancer. This variability presents a major challenge in optimizing treatment strategies and improving patient survival.

The untimely passing of Dr. Mikhail “Misha” Blagosklonny has left a lasting void in geroscience and oncology. This review examines his profound contributions, focusing on his pioneering the Hyperfunction Theory and his advocacy for rapamycin, an mTOR inhibitor, as a therapeutic agent for lifespan extension.

Sleep is essential for everyone, but for those living with dementia, it is vital for better health and quality of life. Addressing sleep problems in dementia care is a crucial step toward improving life for both patients and caregivers.